|
1/15/2024 0 Comments Charge of carbon in knac4h4o64h2oThe atomic radius of a chemical element is a measure of the distance out to which the electron cloud extends from the nucleus. It must be noted, atoms lack a well-defined outer boundary. The atomic radius of Carbon atom is 69pm (covalent radius). Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally-occuring isotopes and their abundance. 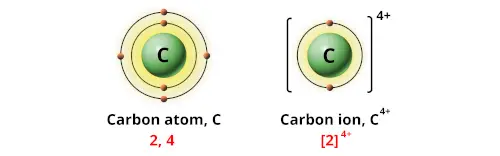
The atomic mass is carried by the atomic nucleus, which occupies only about 10 -12 of the total volume of the atom or less, but it contains all the positive charge and at least 99.95% of the total mass of the atom. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Mass numbers of typical isotopes of Carbon are 12 13. 
Isotopes are nuclides that have the same atomic number and are therefore the same element, but differ in the number of neutrons. The difference between the neutron number and the atomic number is known as the neutron excess: D = N – Z = A – 2Z.įor stable elements, there is usually a variety of stable isotopes. Neutron number plus atomic number equals atomic mass number: N+Z=A. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol N. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10 -19 coulombs. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. 】dots or two valence electrons.Atomic Number – Protons, Electrons and Neutrons in CarbonĬarbon is a chemical element with atomic number 6 which means there are 6 protons in its nucleus.Now we need to connect the outer atoms with the central atoms using single bond or line(-).Every line represent two 【 Now we have to determine the central atom in CH₄(methane).The central atom is that kind of atom that is single or that has lower electronegativity.In case of CH₄(methane),Carbon,C, is the central atom and H is the outer atom. So, CH₄ (methane) has totally eight balence electrons( 4+4=8 ) Now we have to count total number of valence electrons for CH₄(methane). The nunber of valence electrons in hydrogen is 1.ĬH4 Lewis structure Setup Step-2: CH4 Valence Electrons The highest value of principal quantum number here is n=1. To get the valence electrons of hydrogen,we need to look at the electronic configuration of hydrogen. We know a square has four sides.At first,we have to place one valence electrons as dot to every side of that square before pairing up.Īgain, we have to find out the valence electrons of hydrogen. Now,to draw the lewis dot structure of carbon,we need to imagine a square around carbon. The nunber of valence electrons in carbon is 4. The highest value of principal quantum number ,n, indicates the valence shell and we know the electrons in valence shell is called valence shell. 
The highest value of principal quantum number here is n=2. To get the valence electrons of carbon,we need to look at the electronic configuration of carbon. To draw the CH4 Lewis structure, we have to find out the CH4 valence electrons first.We express valence electrons as dots in lewis dot structure.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
